A 58-year-old former engineer with stage four lung cancer has been driven to place his Kajang home on the market in a desperate attempt to fund the treatment keeping him alive, as the punishing cost of immunotherapy forces him to weigh survival against losing the roof over his family’s head.
“I have already put up my house for sale, but have not gotten a buyer. I need money for my treatment,” he said, describing how he advertised the property before Deepavali last year, only to face six agonising months without success.
“I need financial help… I need to be around for my family. I have responsibilities. I cannot leave them hanging. I am holding on to my life for them only,” he added.
Malaysiakini is withholding the man’s identity at his request, as two of his family members are government servants and he fears their jobs would be affected.
His ordeal began when two lumps were first detected on his buttocks, prompting scans that revealed advanced lung cancer and a large mass near his heart.
After extensive bloodwork was sent to Singapore, doctors confirmed he required a combination of chemotherapy and immunotherapy.
The costly but effective cancer immunotherapy drug pembrolizumab, sold under the brand name Keytruda from Merck Sharp & Dohme (MSD), was the recommended treatment for his condition.
Already reeling from the shock of a terminal diagnosis, he said he entered the process confused and frightened, with little understanding of what lay ahead.
Though the drug proved remarkably effective, reducing his lung mass from 5.1cm to 3.6cm after just three cycles, the treatment came at a devastating cost of RM17,000 per cycle. Within months, the financial burden became overwhelming.
“Three cycles cost me RM51,000 already. One cycle is already RM17,000. All my savings are gone,” he said in a phone interview.
Having already spent over RM100,000 on heart and cancer treatments after a previous heart attack and stroke, he lost his insurance after becoming unemployed and could only depend on dwindling savings.
Pembrolizumab is approved for the treatment of more than 20 types of cancer worldwide, making it one of the most widely used immunotherapy drugs in modern oncology.
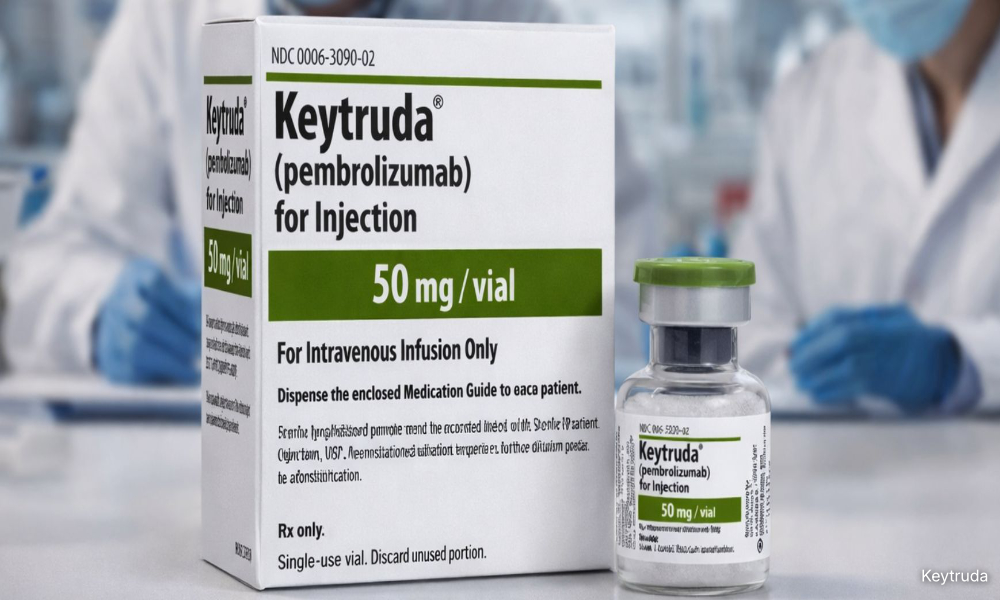
Its approved uses span a broad range of cancers, including lung cancer, melanoma, head and neck cancers, bladder cancer, kidney cancer, cervical cancer, breast cancer, colorectal cancer, liver cancer, and certain blood cancers.
The medicine is also used in some tumour-agnostic cases, where treatment is based on specific genetic or biomarker characteristics rather than the cancer’s location in the body.
However, patient eligibility depends on the type and stage of cancer, as well as regulatory approvals and clinical guidelines in individual countries.
In Malaysia, the drug is marketed through MSD (Malaysia) Sdn Bhd, while distribution is often handled by Zuellig Pharma Sdn Bhd.
Confusion over pricing and limited explanations
At a private hospital, where the cancer treatment programme was being carried out, the patient described the experience of treatment planning and communication as unclear and incomplete.
He said when pembrolizumab and chemotherapy were first introduced, there was “no complete plan for a cancer patient”, adding that this was “a lacking point from the hospital”.
“Basically, no knowledge for people like us. We were ‘blur’,” he said, alleging that he was not properly guided through the process.
He explained that although he was initially considered for a pre-trial medicine programme, he was found to be ineligible due to the timing of a previous heart attack.
The programme, as he understood it, would have fully covered treatment costs but carried a risk of death if the medicine did not work. He ultimately did not proceed.
“They never explained how this works, what is the plan, etc,” he said, referring to the cancer treatment programme, adding that he was only given a pamphlet.
He said he was also told he could apply to the National Cancer Society Malaysia (NCSM) for assistance after completing a number of treatment cycles, but that he was not properly informed about the requirements.

He also later learned that he would still need to pay for about nine to 10 cycles before he would be eligible for assistance.
“I did not know any of this. I was totally blank, with cancer, and a proper explanation was not given.”
He also described uncertainty around cost and supply. While he was told each cycle cost RM17,000, he later questioned the pricing and said he believed the drug was available elsewhere at a lower rate.
“I just want to know, what is the actual price of this medicine?”
He also said he was unclear about how the medication was prepared and administered, noting he was unsure whether he received the full dosage as stated.
Despite these concerns, he acknowledged the treatment had a clear clinical effect, saying the mass in his lung reduced significantly from 5.1cm to 3.6cm within three cycles.
Overall, he described the experience as confusing and emotionally and financially difficult, stating that he felt he had to navigate much of the process himself while dealing with uncertainty about both treatment and cost.
Once a project manager and family breadwinner, he now finds himself wheelchair-bound as cancer has spread to his leg bone, while his wife and children shoulder mounting emotional and financial strain.
“They are terribly affected. I was the reliable breadwinner, but now, all falls on my wife and kids,” he said.

Despite his wife being a government servant, he said attempts to secure additional financial support were repeatedly rejected.
“My wife is a government servant. When we asked the doctor in charge to sign the GL (a guarantee letter stating that civil servants and their close family members are eligible for financial aid for medical treatments), he refused, saying government guarantees do not cover pembrolizumab.
“But my justification was, give me a fighting chance. I want to work something out. We approached the same doctor two to three times, but he refused. If the government don’t want to give, let them give me a letter then. I can maybe go to politicians and request help,” he lamented.
Appeals to charities and aid organisations also yielded little meaningful help. Forced to stop immunotherapy because he could no longer afford it, he watched in despair as his tumour began growing once more.
“After I stopped immunotherapy and just stuck to chemotherapy, my mass grew to 4.1cm after my CT scan. So, I knew immunotherapy worked for me and is the solution,” he said.
Now, facing the brutal reality that the treatment prolonging his life may remain financially out of reach, he is appealing to public generosity, hoping even modest donations can help him continue.
“If these medicines are good, why can’t the government offer them for free? Not asking for all patients, but for eligible ones,” he said, questioning why access to potentially life-saving treatment should come at such catastrophic personal cost.
Call for a more holistic approach
NCSM managing director Dr M Murallitharan said when cheaper versions of a drug enter the market, it can dramatically improve access for patients by driving prices down across the board.
Using another cancer treatment drug, trastuzumab, as an example, he explained that it originally cost a five-digit amount when first introduced. However, once generic or biosimilar drugs became available, even the price of the original branded drug fell significantly, first into the four-digit range, and eventually close to the three-digit range.
“(For) most cancers, waiting 15 to 20 years for generics to come is a long time, because of patenting and related issues. This brings us back to questions about pricing.
“Pharmaceutical companies do argue, quite rightly, that they invest heavily in innovation, and that only one in a hundred experimental drugs ever reach the market.
“However, having said that, I think pharmaceuticals also have a duty; when bringing a new drug to market, to consider how we ensure a sustainable model for their revenues.
“What we are increasingly seeing is that pharmaceuticals are focused on return on investment (ROI) within two to three years.
“Instead of that, there should be a focus on long-term returns. Long-term ROI would mean that upfront prices are not so high. Both pharmaceuticals and regulators need to start thinking in this way,” Murallitharan told Malaysiakini.

He said that while some patients do end up selling their homes to fund treatment, this is often not a straightforward solution because property cannot be quickly liquidated.
As a result, many patients resort to other forms of financing, including taking bank loans, mortgaging their homes, or, in some cases, turning to high-interest personal loans and even loan sharks.
“Keytruda is a recurring cost. That means patients often have to rely on crowdfunding every few months, repeatedly. In that sense, it doesn’t really work as a sustainable solution, and with so many patients needing Keytruda from time to time, it becomes very difficult to continually raise funds for it,” he added.
Murallitharan said he has also helped patients who come to NCSM for aid using his own funds.
“I think I’ve already explained quite clearly how NCSM is playing its part. Also, Cancer Research Malaysia (another organisation) is not a funding organisation; it is a research body, so it does not provide patients with access to treatment,” he added.
He then referred to the National Cancer Council (Makna), a cancer support charity, saying it is also financially constrained.
“Makna does support some patients, but even then, it is facing significant funding challenges, and it has never funded Keytruda. Keytruda is simply too expensive for it to cover,” he added.
MSD and Zuellig respond
Malaysiakini has also contacted the two pharmaceutical companies and the private hospital where the patient received treatment.
The name of the private hospital is being withheld pending its response.
Zuellig refused to weigh in, saying it only supports product distribution and patient programme administration in Malaysia.
“Therefore, we do not provide comments on behalf of an external party regarding this matter,” said the company’s representative via email.
MSD, meanwhile, issued a lengthy statement in response to queries from several media organisations.
“MSD typically works with major international distributors outside the US to leverage local market expertise in managing sales, pricing, inventory, and distribution of our medicines and vaccines.
“Regarding patient access in Malaysia, for those who need support, a financial patient programme is available and administered through the National Cancer Society of Malaysia (NCSM) as the service provider,” said MSD senior vice-president and chief communications officer, Johanna Herrmann.
She added that MSD is dedicated to making its medicines available to patients who may benefit from them as soon as possible, adding that this is why it invests in the continued development of its drugs and vaccines, and also provides support programmes and services to help ensure that those who are prescribed Keytruda have access to the company’s medicine.
“Our medicines and vaccines are priced differentially across markets, and sometimes within markets, according to numerous factors. These factors include the value a therapy brings to patients and the healthcare system, countries’ pricing and reimbursement systems, and the ability of governments to finance healthcare.
“We recognise the challenges with traditional access models and the increasing political and business pressures on access, pricing, and IP from emerging markets.
“MSD actively works in partnership with various stakeholders, including private enterprises, government agencies, multilateral and non-governmental organisations, to help ensure our science advances health care. These collaborations are a key part of our work to enable access to healthcare that is affordable, efficient, equitable, and sustainable on a global scale,” she added.
Herrmann said that when market-based solutions are inadequate or unavailable, MSD pursues programmes that provide direct access to its medicines and vaccines, including product donations and patient assistance programmes.
“We strongly reject any assertions that suggest we have fallen short of the high standards of transparency and integrity that govern how we deliver care to patients,” she added.
The questions were sent via the International Consortium of Investigative Journalists (ICIJ), which embarked on a year-long investigation of the world’s healthcare system in which pharmaceutical companies like MSD allegedly receive favourable treatment and use patents, prices, and regulatory loopholes to push costs higher, while allegedly putting cancer patients into debt.

The investigation examined how one of the world’s largest drugmakers deployed tactics to inflate the volume of prescriptions and keep prices high through lobbying and seeking to delay cheaper versions of the drug from reaching hundreds of thousands of cancer patients in the coming years.
ICIJ claimed that this is playing out as governments around the world spend growing amounts on Keytruda, with steep prices straining government budgets, even in wealthy countries.
Prices for a year’s treatment range from about US$80,000 (RM317,000) in Germany, US$208,000 (RM825,600) in the US, US$93,000 (RM369,000) in Lebanon, US$130,000 (RM516,000) in Colombia, US$65,000 (RM258,000) in South Africa, and US$116,000 (RM460,000) in Croatia.
Malaysiakini is part of this investigative programme, which brought together journalists from five continents. For the report, Malaysiakini examined the situation in Malaysia.
Cancer-related claims increase
The Social Security Organisation (Socso) said cancer-related claims in the last five years have increased at a compound annual growth rate (CAGR) of 4.94 percent.
However, it added that the exponential increase in 2023 was driven by adjustments to the cost of living for invalidity-pension payouts.
“Collectively from 2021 to November 2025, Socso has received 21,214 reported cases related to cancer, of which 5,492 cases were approved by the Medical Board, representing an approval rate of approximately 26 percent.
“The rejected claims of invalidity due to cancer, provided they have fulfilled their contribution entitlement, are mostly due to early-stage cancer diagnosis and have not achieved invalidity upon Medical Board examinations.
“With modern mainstream cancer treatment, the prognosis of cancer patients has improved, hence significant loss of functions or earning capacity may be avoided to result in invalidity,” said Socso CEO Dr Azman Aziz Mohammed to Malaysiakini.
He said breast cancer, colorectal cancer, and cervical cancer are among the top three cancers in Malaysia. However, he explained that these have good screening tools with high specificity and sensitivity, such as mammograms, stool occult blood tests, and pap smears.

He said Socso is a strong advocate for early cancer screening, hence it is included in its Health Screening Programme (HSP), free of charge for those who meet the requirements and are over 40.
“However, only 45 percent of women underwent a pap smear test, and five percent of women performed a mammogram among 20,000 women screened last year. The complications of late-stage cancers may be avoided with early action, and the importance of screening couldn’t be more highlighted with these low uptakes,” he explained.
When asked if Socso has reviewed claims involving patients prescribed high-cost cancer drugs such as pembrolizumab and the outcomes for such patients, Azman said no study has been done specifically on patients for treatment potency and prognosis.
“Not mandated by our act,” he added.
Azman said that though there has been a gradual and sustained increase in cancer-related claims, this is linked to broader health trends, rather than a single cause.
He said that claims related to non-communicable diseases (NCDs) have also risen by about 4.15 percent over the past five years, while overall invalidity claims have increased by 6.08 percent in the same period. He suggested that the rise in cancer claims is likely part of this wider upward trend in chronic diseases.
He added that as NCDs become more common, cancer-related claims are expected to continue increasing steadily. - Mkini




No comments:
Post a Comment
Note: Only a member of this blog may post a comment.